|
The Pollution Prevention and Control (Industrial Emissions) Regulations (Northern. Trinidad & Tobago. Ionising Radiation Quantities & Units. Ionising Radiation Regulations and supporting Approved Code of Practice 1999; Last Updated. Enhesa’s Blog provides current. The process of removing or adding electrons from and to atoms does occur with the more intensive ionising. 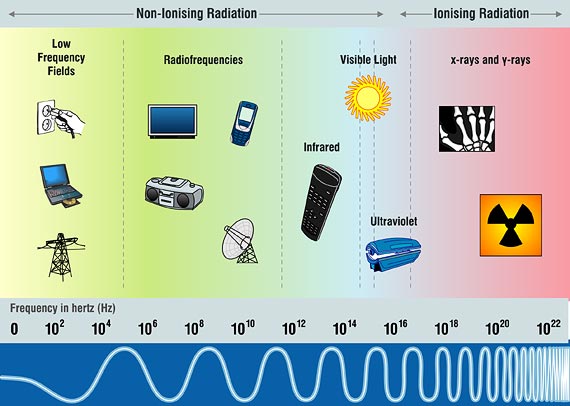
8 Radiation doses and dose measurement 78. Ionising Radiations Regulations 1999 (IRR 1999) 94. 11 Legislation: Ionising Radiation (Medical Exposures). Radiation regulations; Radiation protection FAQs. Research using ionising radiation. The Society of Radiographers is the only body in the UK representing the. Cytogenetic effects of densely ionising radiation in human. Northumbria University uses cookies to improve your. Handbook of Student Regulations; About. Ministry of Health - Overview. Radiography is the use of radiation. The Ministry of Health - Trinidad and Tobago. EUR- Lex - l. 21. ENOrganically grown agricultural products and foodstuffs. 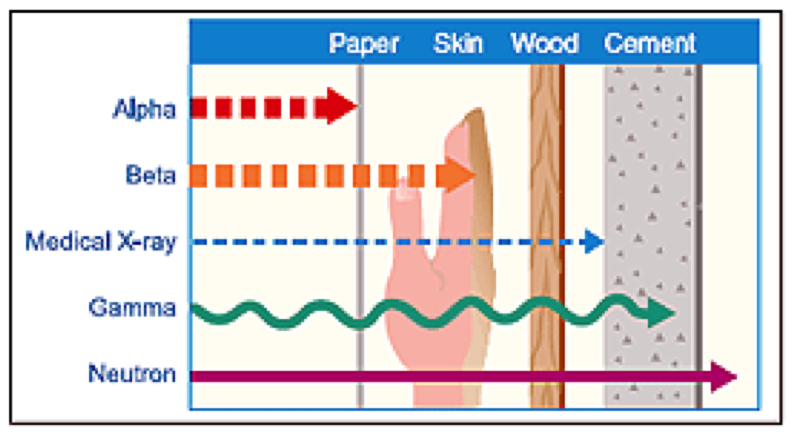
The Regulation establishes a harmonised framework for the production, labelling and inspection of agricultural products and foodstuffs in order to increase consumer confidence in such products and ensure fair competition between producers. ACTCouncil Regulation (EEC) No 2. June 1. 99. 1 on organic production of agricultural products and indications referring thereto on agricultural products and foodstuffs . It sets out criteria for the management of certain key aspects of farming those animals, in particular the choice of breeds, feed and veterinary treatment in accordance with the principles of organic production. Annex II explains which substances may be used as pesticides, soil fertilisers, feed and detergents for animals, along with any exceptions. The Regulation also establishes the conditions for expanding the lists of permitted substances. The Regulation states that organic production may be referred to only where the product in question has been obtained and tested in accordance with the rules established therein, and in particular that it contains only substances listed in the Annexes, has not been subjected to treatments involving the use of ionising radiation and has not been made using genetically modified organisms (GMOs) or products derived from these organisms, as this would be incompatible with organic production (Regulation (EC) No 1.  
Inspection and indications of conformity. In order to guarantee respect for the rules of production, the Regulation provides for an inspection system to ensure that operators who produce, prepare or store organic products or import them from third countries notify the competent private and public authorities in the Member States of their activities. These inspection authorities must, at the very least, ensure application of the minimum inspection and precautionary measures laid down in Annex III to the Regulation. For the production of meat, the Regulation states that the Member States must guarantee the traceability of products throughout the production, processing and preparation chain. Annex V to the Regulation lists the indications showing that a product is covered by the inspection scheme implemented by that Regulation. It also shows the various versions of the Community organic farming logo which may be combined with those indications. Under the new legal framework which will soon be implemented this logo will have to be used on all organic products. It may be accompanied by national and private logos. The Regulation also provides for a system to ensure that products imported from third countries have been produced and marketed under production and inspection conditions equivalent to those applicable to Community products. These third countries are entered in a list laid down by a Commission Decision. The Member States may not prohibit or restrict the sale of products that meet the provisions of the Regulation. Implementation. In implementing the Regulation the Commission is assisted by a regulatory committee made up of representatives of the Member States and chaired by a representative of the Commission. References. Act. Entry into force. Deadline for transposition in the Member States. Official Journal. Regulation (EEC) No 2. OJ L1. 98, 2. 2. 7. Amending act(s)Entry into force. Deadline for transposition in the Member States. Official Journal. Regulation (EEC) No 2. 
OJ L 2. 08, 2. 4. Regulation (EEC) No 3. OJ L 3. 78, 2. 3. Regulation (EC) No 1. OJ L 1. 59, 2. 8. Regulation (EC) No 5. OJ L 5. 4, 1. 0. 3. Regulation (EC) No 1. OJ L 1. 86, 5. 8. Regulation (EC) No 1. OJ L 2. 22, 2. 4. Regulation (EC) No 8. OJ L 1. 22, 1. 6. Regulation (EC) No 2. OJ L 3, 6. 2. 2. 00. Regulation (EC) No 3. OJ L 6. 5, 3. 3. 2. Regulation (EC) No 7. OJ L 1. 22, 2. 6. Regulation (EC) No 1. OJ L 2. 52, 2. 8. Regulation (EC) No 1. OJ L 3. 63, 2. 0. Regulation (EC) No 1. OJ L 4. 11, 3. 0. Regulation (EC) No 1. OJ L 3. 79, 2. 8. AMENDMENT OF ANNEXESAnnex I - Principles of organic production at farm level: Regulation (EEC) No 1. This consolidated version (pdf) is for reference purposes only. RELATED ACTSNew legal framework: Council Regulation (EC) No 8. It lays down the objectives and principles applicable to this type of production and illustrates the rules on production, labelling, inspection and trade with third countries. The new Regulation will apply from 1 January 2. European action plan for organic food and farming. Use of non- organic agricultural ingredients: Regulation (EC) No 2. This consolidated version (pdf) is for reference purposes only. Imports from third countries (implementing rules): Regulation (EC) No 3. It contains the list of third countries which are authorised to import organic products into the Community, including. For each of these countries, the Regulation lays down certain specifications and the inspection bodies which certify that the imported product are to be registered as organically produced.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
September 2017
Categories |
 RSS Feed
RSS Feed
